
Nuclear fusion; It is the process that gives the sun its life, yet it also inevitably destroys entire solar systems. We owe our thanks to this phenomenon of physics for most of the elements on the Periodic Table (at least up to uranium), and it has the potential to unlock a more eco-friendly source of energy that may very well outperform and surpass solar panels. But unfortunately, a quantum-sized wall (literally) stands in our way to this Pandora’s Box of power.
While nuclear fusion is hardly mentioned in Star Trek, it is often referenced in science fiction in general. Star Trek has, unfortunately, overlooked one of the greatest undertakings of modern technology in lieu of more exotic sources of energy (anything that sounds fancy and high-tech); nuclear fusion may very well be the next environmentally-safe alternative to windmills. One of the greatest gifts of nuclear fusion is the promise of an “everlasting” supply of energy.
In this installment of “Science Fiction or Science Fact,” we’ll examine the physics of nuclear fusion in order to understand and appreciate why it matters so much and finally asses its scientific applicability.
The Breakdown
In the grand scheme of energy production, the process of generating energy is only as useful as the amount of net energy you produce at the “end of the day.” Or rather that the amount of energy one harnesses from a reactor is greater than the amount required to maintain it. To put this into some perspective, energy production is analogous to day trading in the stock market: stocks are bought when they are cheap and sold later in the day when they increase in value; the result is a net gain in profits. Energy production works the same way—except harnessing power from thermodynamic processes is less risky than owning stocks and it is much more predictable and calculable.
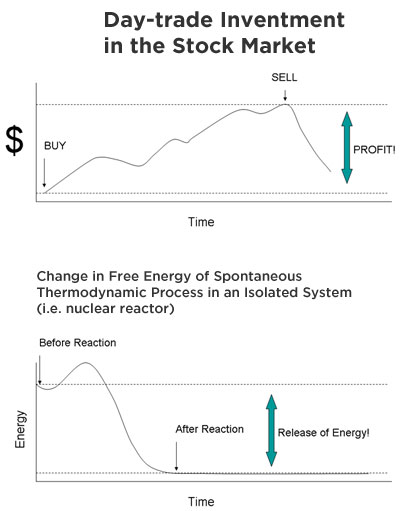
You may recall one of my earlier posts about antimatter reactors and how society cannot become reliant on power plants that consume more energy than it produces (though it may be sufficient to power warp engines, but that’s beside the point). What makes nuclear fusion so special is its greater return in energy.
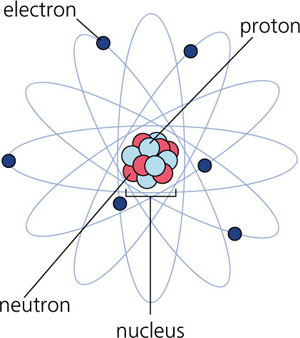 Atoms are one of the smallest particles known to exist (with the exception of elementary particles). Atoms are made up of a nucleus containing protons and neutrons: protons are positively-charged while neutrons have no charge. In culmination, the nucleus is the positively-charged core of the atom, while the electrons that “orbit” around the nucleus (as presented in the atomic model) are negatively-charged. The model presented here isn’t entirely inaccurate, though the atom is drawn hopelessly out of scale: if an atom was as large as a football stadium, then the nucleus would be no larger than a grain of rice at the very center of the field.
Atoms are one of the smallest particles known to exist (with the exception of elementary particles). Atoms are made up of a nucleus containing protons and neutrons: protons are positively-charged while neutrons have no charge. In culmination, the nucleus is the positively-charged core of the atom, while the electrons that “orbit” around the nucleus (as presented in the atomic model) are negatively-charged. The model presented here isn’t entirely inaccurate, though the atom is drawn hopelessly out of scale: if an atom was as large as a football stadium, then the nucleus would be no larger than a grain of rice at the very center of the field.
The reader may question the stability of the atomic nucleus. If it is composed of positively-charged protons, how can the nucleus remain intact without the positive charges repelling each other? The answer: the Strong Nuclear Force (the strongest of the four fundamental forces known to exist in the Universe). While the like-charges of protons repel one another, it is the Strong Nuclear Force that abrogates any repulsive interaction between protons and tightly binds them together.
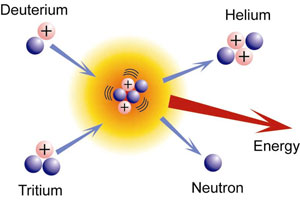 The Strong Nuclear Force is what finally drives the fusion of two nuclei into one. However, it only works at the subatomic scale and cannot reach out and pull nuclei in toward each other to fuse them together. This is why extreme momentum is required to initiate the fusion reaction; two nuclei must be traveling so quickly that they overcome any electrostatic repulsion, collide into one another, and fuse. Inside stars, heat and pressure collide tritium and deuterium (isotopes of hydrogen) into one another to cause the two nuclei to fuse into helium, expelling an extra neutron.
The Strong Nuclear Force is what finally drives the fusion of two nuclei into one. However, it only works at the subatomic scale and cannot reach out and pull nuclei in toward each other to fuse them together. This is why extreme momentum is required to initiate the fusion reaction; two nuclei must be traveling so quickly that they overcome any electrostatic repulsion, collide into one another, and fuse. Inside stars, heat and pressure collide tritium and deuterium (isotopes of hydrogen) into one another to cause the two nuclei to fuse into helium, expelling an extra neutron.
Another interesting note is that the mass of helium plus the mass of the expelled neutron is found to be lighter than the original atoms that were mashed together to form the product. So where did the extra mass go? The force of the impact and the Strong Nuclear Force that resolves the stability of the helium nucleus literally converted some of the matter into its mass-equivalent of pure energy. That is A LOT of energy…imagine the power output of trillions upon trillions of nuclei fusing together at once!
In summary, nuclear fusion occurs when intense pressure causes two nuclei to collide and smash into one another and fuses them into a larger nucleus, a process that liberates a remarkable amount of energy. Here is a video that breaks it down step-by-step:
Nuclear fusion is the reason why the sun shines so brightly and so hotly; it is also the reason why scientists regard nuclear fusion as the “Holy Grail” of energy because the net gain in power would solve just about any economic and environmental problem on Earth for millennia. Imagine the benefits it would have: electricity bills might disappear, cars could run on and on without ever refueling, salt water could be easily desalinated for communities with limited access to clean, drinking water, global climate change becomes a nonissue, societies become self-sufficient, and so much more. This is the promise of nuclear fusion (sun panels and even nuclear fission would be thrown out the window for this extraordinary power source). The best part is: it’s just around the corner!
The Final Verdict
Nuclear fusion isn’t just possible, but it occurs in the sun, it has been generated in hydrogen bombs, and it may very well happen in controlled laboratory experiments because nuclear fusion is a Science Fact. The only obstacle in our way is reproducing a sustainable, nuclear reaction in the lab for the sole purpose of harnessing energy from it.
I cannot stress enough the “gravity” (excuse the pun, if you know what I mean) and difficulty of nuclear fusion. It requires heat and pressure on the scales found in stars; as such scientists cannot hope to recreate whole suns just to run a nuclear fusion reactor. Our next best hope is to find a circumvention of physics that allows us to bypass the electrostatic repulsions of nuclei and force them to fuse spontaneously.
Well…here it is:
Two hundred high-intensity lasers focused on a single casing of deuterium and tritium may be the key to unlocking the power of the sun for our benefit. It may not become marketable in the near future, but scientists and engineers wouldn’t be working diligently on it if it wasn’t possible.
Tom Caldwell has a Bachelor’s of Science in biochemistry from UCLA. He is currently working towards a Ph.D. in molecular biology.




























































Dean Holyer
September 17, 2011 at 6:56 am
Yesterday while I was surfing through the U Tube videos, I watched a collection of videos on Fusion Energy Production (FEP Projects). Most came from the 2007 to 2010 years and most dealt with Returning to the Moon with the NASA Constellation Project. These film clips stressed facts of over 4 billion years of He3 on the Moons surface. The He3 would be skimmed off the surface and sent back to Earth and valued at $3Billion per ounce or pound as He3 is a clean fuel to mix with Deuterium in Fusion Reactors. The Deuterium/Tritium Fusion reaction creates a High Energy Neutron in the process witch is Radioactive and destructive and dangerous. The Deuterium/Helium3 reaction only creates a High Energy Proton that is almost as dangerous as the Neutron but has a Positive charge so it can be controlled as simple as the beam used in 20th Century Picture tube televisions. A High energy and velocity Neutron is almost as elusive as a beam of light and mirrors were never invented.
Some of the video creators/authors even valued the Lockheed Martian X-33 Venture Star as a replacement for the NASA Space shuttle Program. This spaceship was very similar in purpose as the STS Space Shuttle but is a SSTO (Single Stage To Orbit Vessel) with it’s Vector Trust type Rocket Engines, a higher velocity steerable Vectored trust exhaust engine, and could fly similar to commercial aviation planes with an internal non disposable fuel storage supply that could use any International Airport to launch from and land at. And with fuel storage to refuel the tanks of the Venture Star. This spaceship could travel to Lunar orbit, Mars or even to Asteroids.
The X-33 has a cargo bay of around 2/3rds the volume of the Space Shuttle, but could have a crew of personnel as large as 10 and if the cargo bay was partly used for food and air storage the Venture Star could remain in Space for Months.
This could become possible with the replacement of America’s current President, And NASA re-devoted to the Moon then Mars and beyond. NASA may still land on Mars first in 2035, whuch is only 2 years past the plans that erased our return to the Moon by 2019. From 2020 to 2030 America would be colonizing the Moon and by 2035 missions to land and stay on the surface of Mars may be ventured upon.
As we venture to Mars, the visitors may not be government funded or sent personal, but may be Lunar residents seeking new frontiers to explore and inhabit under total personal freedom. The big question is whom will be first to declare indendant freedom first, the Moon residents.
Some of my personal knowledge has been given to me by my brother who is a Mars Exploration Engineer whom has worked on many of the robotic projects sent to Mars since he started working there in 1983 at the Denver/Waterton Canyon building that use to be the Martian Marietta plant that the Mars Viking lander’s from America’s bicentennial year and Colorado’s Centenial year.
I have even herd rumors of future ventures by Lockheed Martian Colonizing and inhabiting the Asteroids after Mars in 2040 and about every 10 years a planet is reached. And by the start of the 22nd Century we may be reaching the Ort Cloud that in cases the Sol Solar System, and starting to have dreams of reaching for and exploring the Stars.
Who knows when the next Century is reached, ION thrusters may have evolved into high velosity engines or we may have actually started work on Anti Matter energy sources or thrusters, may be our Atomic structure understanding will have showed us Earthlings how to warp Time/Space to give us Warp Drive or we may still be limited a percentage of the speed limit of light. But that is 90 years or more from now. But those dreams are still dreams, it is almost like asking the Right brothers after there first flight to start booking flights to Austrailia for the 1920 Christmas holiday